Can Low Vitamin D Cause Fatty Liver
- Original Research Article
- Open Access
- Published:
The association between vitamin D status and non-alcoholic fatty liver disease in adults: a hospital-based study
- Mohamed Rezk Elmedames1,
- Ayman Ramadan Abdelhai1 &
- Ayman Mohamed Marei2
Egyptian Liver Journal volume 10, Article number:25 (2020) Cite this article
-
3966 Accesses
-
2 Citations
-
Metrics details
Abstract
Background
The association between vitamin D levels and non-alcoholic fatty liver disease (NAFLD) has been increasingly determined in recent researches. The aim of this study was to determine the association between vitamin D levels, measured as serum 25-hydroxy vitamin D (25(OH)D) and NAFLD. Serum 25(OH)D was prospectively determined in 80 patients. Of them, 40 subjects had NAFLD, whereas 40 subjects served as healthy control. Vitamin D deficiency was defined as serum 25(OH)D level < 20 ng/ml. Receiver operating characteristic (ROC) and regression analysis methods were used in our analysis.
Results
About 70% of patients with NAFLD had vitamin D deficiency, but only 35% in the control group had. The mean serum 25 (OH)D was significantly lower in patients with NAFLD than the healthy control group (16.13 ± 10.23 versus 27.35 ± 10.58 ng/mL; P < 0.001). ROC curve analysis revealed that serum 25(OH)D level of less than 11.2 ng/ml increases the risk of NAFLD with 45% sensitivity and 97.5% specificity (Serum 25(OH)D level ≤ 18.1 ng/ml in males and ≤ 9.4 ng/ml in females increase the risk of NAFLD). Multivariate regression analysis showed that vitamin D deficiency, high age, and high BMI were associated with a significant high risk of NAFLD.
Conclusion
NAFLD patients have low serum vitamin D concentrations, suggesting that vitamin D may have a role in the development of NAFLD. Future studies are recommended to determine the important therapeutic implications of vitamin D for the prophylaxis or the treatment of NAFLD.
Background
Non-alcoholic fatty liver disease (NAFLD) is a clinico-histopathological entity with histological features that occurs in patients with little or no history of alcohol consumption. Patients with NAFLD have hepatic steatosis, with or without inflammation and fibrosis [1]. NAFLD is classified into non-alcoholic fatty liver (NAFL) and non-alcoholic steatohepatitis (NASH). In NAFL, hepatic steatosis is present without evidence of inflammation; while in NASH, hepatic steatosis is associated with hepatic inflammation [2].
The multiple-hit hypothesis is better framing the variable circumstances implicating the development and progression of NAFLD [3]. However, recent studies show that the progression of NAFLD is not always linear, it is not who is more likely to transform into more advanced stages [4], considered as the hepatic manifestation of NAFLD in most cases associated with type 2 diabetes and dyslipidemia [5].
Fatty liver is the hepatic component of metabolic syndrome. The American Heart Association/National Heart, Lung, and Blood Institute and the International Diabetes Federation considered a patient as having metabolic syndrome if he has at least three abnormal findings of the following: abdominal obesity, low high-density lipoprotein, cholesterol, elevated blood pressure, high fasting glucose, or high triglyceride concentration [6].
Vitamin D has been associated with many disease pathogenesis including autoimmune disease, cardiovascular disease, cancers, inflammatory processes, and liver diseases [7,8,9]. The association between vitamin D levels and NAFLD has been increasingly determined in recent researches [9].
The pathogenesis of the association between NAFLD and low vitamin D levels is still undetermined; however, protective anti-fibrotic and anti-inflammatory function of vitamin D on the hepatic stellate cells has been suggested [10]. Vitamin D reduces free fatty acid-induced insulin resistance in peripheral tissues and in hepatocytes [11]. Therefore, low vitamin D level may lead to intrahepatic lipid accumulation which is responsible for NAFLD pathogenesis [12]. Abramovitch and colleagues confirmed the anti-fibrotic effects of vitamin D through inhibition of hepatic stellate cells proliferation in an in vivo murine model [12].
NHANES III database reviewed more than 6000 patients; of them, about 300 patients showed unexplained elevation in alanine aminotransferase (ALT) and lower vitamin D levels than the control group [13].
Given that vitamin D deficiency and NAFLD have direct and indirect associations with obesity and sedentary lifestyle, it is not unexpected that vitamin D deficiency would be a co-factor in the pathogenesis of NAFLD. Therefore, the aim of this study was to determine the relationship between the serum vitamin D level assessed by 25-hydroxyvitamin D3 (25(OH)D3) and NAFLD in adults.
Methods
Study design
A hospital-based non-interventional cross-sectional controlled study was carried out in the Internal Medicine Department and Immunology Research Lab in Microbiology and Immunology Department at Faculty of Medicine, Zagazig University Hospitals. The institutional review board approved the study (ZU-IRB#3776-30-5-2017). Written informed consent was obtained from all individual participants in the study.
Patients' selection and data collection
Eligible patients had a diagnosis of NAFLD, were aged 18 to 60 years old, and had bright hepatic texture proven with abdominal ultrasound. Matched normal control subjects with normal liver ultrasound /liver chemistry were included. The individual participants in the study were classified into the NAFLD group and healthy control group, were matched in age, gender, residence, the existing comorbidities, and the smoking status.
We excluded patients with chronic viral hepatitis, liver cirrhosis secondary to any cause, primary biliary cirrhosis, primary sclerosing cholangitis, and pregnant women, and patients on calcium or vitamin D supplementation and renal diseases. We also excluded any patients that received vitamin D supply in the previous 2 months.
The following data were collected for each patient eligible for the study: age, gender, residency, smoking status, body mass index (BMI), fasting blood glucose (FBG), hemoglobin A1C (HbA1C), hemoglobin, white blood cells, platelets, total bilirubin, direct bilirubin, alanine transferase (ALT), aspartate transferase (AST), albumin, total plasma protein, alkaline phosphatase, international normalized ratio (INR), creatinine, blood urea nitrogen (BUN), cholesterol, triglyceride, low-density lipoprotein, and high-density lipoprotein. In addition, serum 25(OH)D level was measured for the both groups. Vitamin D deficiency was defined as 25(OH)D levels < 20 ng/ml, while vitamin D insufficiency and sufficiency mean that serum 25(OH)D are equal to 20–30 ng/mL and 30–100 ng/mL, respectively [14,15,16].
Assessment procedures
Blood samples were obtained from each patient and kept for 30 min for spontaneous clotting. Samples were then centrifuged at 3000 rpm for 5 min and stored for up to 5 days at 2 to 8 °C prior to assaying. Specimens held for a longer time (up to 2 months) and were frozen only once at − 20 °C prior to assay. Serum 25(OH)D level was measured by DBC immunoassay of 25(OH)D enzyme-linked immunosorbent assay (ELISA, DBC Diagnostics Biochem Canada: CAN-VD-510) at the Immunology Research Lab in Microbiology and Immunology Department, Zagazig University Hospitals [17]. All other laboratory tests, including liver and renal function tests, and coagulation tests underwent using the routine laboratory testing methods. Liver ultrasonography (US) was performed to study liver echogenicity, size, cirrhotic changes, or other abnormalities. Liver Ultrasonography (US) scanning was performed to assess the degree of steatosis. Transient elastography (Fibroscan) for measurement of controlled attenuation parameter (CAP) and liver stiffness measurements (LSM) were performed by an experienced radiologist who was blinded to the clinical data of the patients [18, 19].
We used ultrasonographic grading of fatty liver [20] as the following:
Grade 0: normal liver echogenicity.
Grade I: diffusely increased hepatic echogenicity but periportal and diaphragmatic echogenicity are still appreciable.
Grade II: diffusely increased hepatic echogenicity obscuring periportal echogenicity but diaphragmatic echogenicity is still appreciable.
Grade III: diffusely increased hepatic echogenicity obscuring periportal as well as diaphragmatic echogenicity.
Statistical analysis
All categorical variables were presented in frequency and percentage, whereas numerical variables were presented with mean ± standard deviation (SD). Comparative analysis and inferential statistics were performed using parametric independent t test that was used for comparison in case of Gaussian distribution, while the Mann-Whitney U test was used for non-Gaussian distribution. For categorical variables, the chi-square test (or Fisher's exact test if appropriate) was used. For all statistical tests, P value ≤ 0.05 was considered statistically significant. Spearman's rank correlation coefficient (Spearman's rho) was calculated for 25(OH)D levels and various study parameters. These analyses were performed using the statistical software program, SPSS, for Windows version 25.0 (SPSS; Chicago, IL, USA). Univariate and multivriate logistic regression analysis were used to determine the predictor variables for the presences of NAFLD.
Receiver operating characteristic (ROC) curve analysis was used to identify optimal cutoff values of 25(OH)D levels with maximum sensitivity and specificity for predicting the NAFLD patients. The best cutoff points were the concentration of serum 25(OH)D at the associated criterion of the highest Youden Index from the ROC analysis using the MedCalc software (MedCalc 10 Software bvba, Ostend, Belgium).
Results
Eighty subjects participated in the present study, of whom 40 subjects had NAFLD, whereas 40 subjects served as healthy control. The demographic and baseline characteristics of the subjects based on the groups are presented in Table 1. The two groups were comparable in all demographic data except the BMI, which was significantly higher in the NAFLD Group (30.8 ± 6.3 kg/m2 versus 34.6 ± 4.3 kg/m2, P = 0.012). The means CAP and LSM were significantly higher in the NAFLD Group (p < 0.001).
Full size table
Table 2 shows the results of all laboratory data in the two groups. Blood urea nitrogen (BUN) was lower in the NAFLD Group (14.1 ± 2.3 mg/dL) than the normal control group (17.7 ± 2.4 mg/dL), p = 0.012.
Full size table
Among the patients with NAFLD, the ultrasonographic grading of fatty liver was as follows: 16 (40%) grade 1, 21 (52.5%) grade 1, and 3 (7.5%) grade 3.
Mean serum 25 (OH)D was significantly lower in the NAFLD group (16.13 ± 10.23 ng/mL) than the control group (27.35 ± 10.58 ng/mL). About 70% of patients with NAFLD had vitamin D deficiency, but only 35% in the control group as seen in Fig. 1.
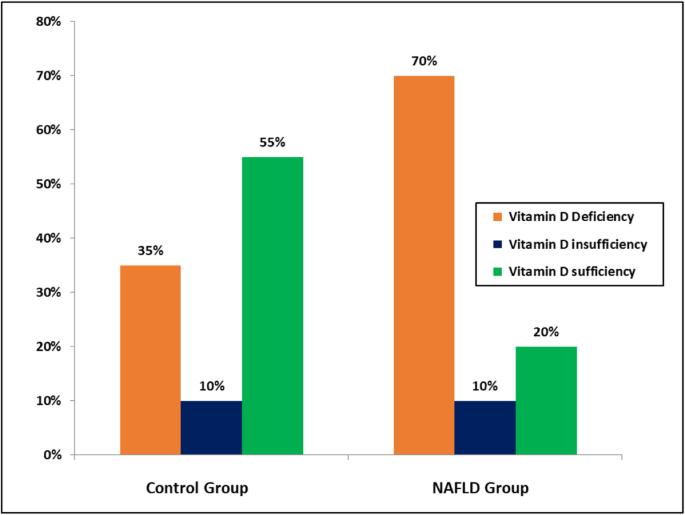
Comparison of the vitamin D status (serum 25(OH)D) between the study groups
Full size image
ROC curve analysis reveals that serum 25(OH)D level of ≤ 11.2 ng/ml can predict the presence of NAFLD with 45% sensitivity and 97.5% specificity (Fig. 2a). Furthermore, in male patients, the cut of level of serum 25(OH)D ≤ 18.1 ng/ml can predict the presence of NAFLD with 76.92% sensitivity and 76.92% specificity (Fig. 2b), while in female patients with 25(OH)D level of ≤ 9.4 ng/ml can predict the presence of NAFLD with 44.4% sensitivity and 99.9% specificity (Fig. 2c). The areas under the curve were 0.78, 0.81, and 0.78 respectively.
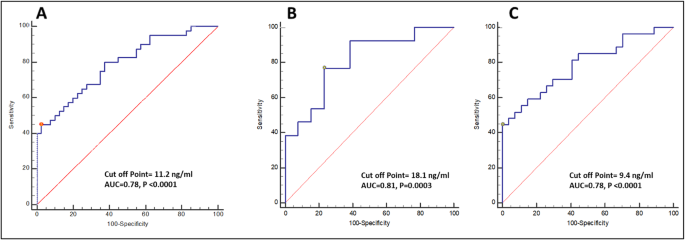
ROC curve analysis of serum 25(OH)D level that can predict the NAFLD in a all patients, b male patients, and c female patients
Full size image
In the univariate logistic regression analysis model, by considering the vitamin D sufficiency as the reference, the risk of NAFLD for the groups with vitamin D deficiency and insufficiency obtained about 5.15 (95% CI 1.78: 14.9) and 2.51 (95% CI 0.46: 13.6), respectively. In addition, patients with high age and BMI were significantly associated with a high risk of NAFLD [OR = 2.08 (95% CI 1.09: 2.17)] and [OR = 2.82 (95% CI 1.71: 5.94)]. In a multivariate logistic regression analysis model, vitamin D deficiency, high age, and high BMI were associated with a significant high risk of NAFLD as presented in Table 3.
Full size table
Discussion
Low vitamin D is prevalent in chronic liver disease patients. Even patients with mild liver disease are affected, although patients with liver cirrhosis are more commonly have severe deficiency [21]. Low serum levels of vitamin D have been observed in chronic liver diseases, especially with liver cirrhosis [8, 22], while in patients with NAFLD, the data are still scanty.
Our study demonstrates that the serum 25 (OH)D was significantly lower in the NAFLD group than the normal control group. More than two thirds of NAFLD patients had vitamin D deficiency, but only one third of the control group had vitamin D deficiency.
The recent systematic review of Pacifico and colleagues [23] included 45 studies exploring the association between vitamin D status and NAFLD/NASH. Of them, 29 studies reported an inverse association between vitamin D status and NAFLD, while 16 studies did not support this association.
On the other hand, the meta-analysis on the relationship between serum vitamin D and NAFLD histologic severity found no association between serum vitamin D levels and disease severity as assessed by NAS and fibrosis score among patients with NAFLD [24].
Using the NHANES III database, Liangpunsakul and Chalasani screened 6800 patients. They found 308 patients with unexplained elevation in ALT and compared their serum vitamin D concentrations with 979 matched controls. Patients with elevated ALT were found to have lower vitamin D levels than the control group, even when controlling for metabolic syndrome and serum triglyceride level [13]. Furthermore, Targher et al. confirmed the association between NAFLD and vitamin D deficiency. In addition, vitamin D concentrations were lower in NASH patients when compared to those with isolated fatty liver [25].
Mean serum 25 (OH)D of 16.13 ± 10.23 ng/mL in the NAFLD group was significantly lower than in the normal control group (27.35 ± 10.58 ng/mL). The results of systematic review [23] showed that the median level of vitamin D in the control group is 27.7 ± 8.75 ng/mL, while the mean level in the NAFLD group is 25.7 ± 8.74.
In the cross-sectional study of Ehrampoush and colleagues [26], the mean serum vitamin D was 26.77 ± 8.26 ng/mL in the healthy group, while in the NAFLD group, it was 15.84 ± 5.50. The results showed that the cutoff point for vitamin D in men and women, vitamin D levels lower than 18 nmol/L for women and 21 nmol/L for men, significantly increases the risk of NAFLD. The ROC curve analysis of the present study reveals that serum 25(OH)D level ≤ 11.2 ng/ml can predict the presence of NAFLD; in male patients, the cut of level of vitamin D ≤ 18.1 ng/ml can predict the presence of NAFLD, while in female patients, it was ≤ 9.4 ng/ml.
In our study, we observed nearly normal levels of liver enzymes in patients with NAFLD. This observation was supported by the recent meta-analysis that conducted to determine the proportion of NAFLD patients with normal ALT value in the overall NAFLD patients [27]. They reported that the proportion of NAFLD patients with normal ALT value in overall NAFLD patients was 25% (95% CI 20–31%).
Many of the epidemiological studies linking low serum vitamin D levels with NAFLD is that the two conditions are associated with obesity (high BMI) [25, 28, 29]. Vitamin D is fat soluble and stored in adipose tissue that increases the sequestration of vitamin D in obese individuals who are also prone to NAFLD [30]. Our study showed that patients with high BMI were significantly associated with a high risk of NAFLD.
The main limitation of our study is the small sample size, which might limit the generalizability of the result. We included only adult's patients, while the study by Manco et al. [31] reported that low levels of 25(OH)D in children with NAFLD correlated with histological severity regardless the metabolic characteristics.
In conclusion, our results showed a significant relationship between vitamin D levels and the risk of NAFLD. Particularly, our study suggests that serum 25(OH)D level less than 9.4 ng/ml for females and 18.1 ng/ml for males can increase the risk of NAFLD. It is important to maintain the appropriate serum level of vitamin D for protecting against NAFLD. Determination of a main role of low vitamin D in NAFLD development might have important therapeutic implications in future studies. In addition, conducting future studies comparing the vitamin D levels in different liver disease is highly recommended. Finally, the association between vitamin D deficiency with prognosis of liver diseases as progression to NASH or development of HCC should be addressed through a prospective studies.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ALT:
-
Alanine aminotransferase
- INR:
-
International normalized ratio
- AST:
-
Aspartate aminotransferase
- BMI:
-
Body mass index
- BUN:
-
Blood urea nitrogen
- FBG:
-
Fasting blood glucose
- HbA1C:
-
Hemoglobin A1C
- NAFLD:
-
Non-alcoholic fatty liver disease
- NASH:
-
Non-alcoholic steatohepatitis
- ROC:
-
Receiver operating characteristic
- SD:
-
Standard deviation
References
- 1.
Younossi ZM, Loomba R, Rinella ME, Bugianesi E, Marchesini G, Neuschwander-Tetri BA, Serfaty L, Negro F, Caldwell SH, Ratziu V, Corey KE, Friedman SL, Abdelmalek MF, Harrison SA, Sanyal AJ, Lavine JE, Mathurin P, Charlton MR, Chalasani NP, Anstee QM, Kowdley KV, George J, Goodman ZD, Lindor K (2018) Current and future therapeutic regimens for nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology 68:361–371
Article Google Scholar
- 2.
Sheth SG, Gordon FD, Chopra S (1997) Nonalcoholic steatohepatitis. Ann Intern Med 126:137–145
CAS Article Google Scholar
- 3.
Buzzetti E, Pinzani M, Tsochatzis EA (2016) The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 65:1038–1048
CAS Article Google Scholar
- 4.
Younossi ZM, Loomba R, Anstee QM, Rinella ME, Bugianesi E, Marchesini G, Neuschwander-Tetri BA, Serfaty L, Negro F, Caldwell SH, Ratziu V, Corey KE, Friedman SL, Abdelmalek MF, Harrison SA, Sanyal AJ, Lavine JE, Mathurin P, Charlton MR, Goodman ZD, Chalasani NP, Kowdley KV, George J, Lindor K (2018) Diagnostic modalities for nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and associated fibrosis. Hepatology 68:349–360
Article Google Scholar
- 5.
Angulo P (2008) Obesity and nonalcoholic fatty liver disease. Nutr Rev 65:S57–S63
Article Google Scholar
- 6.
Gastaldelli A (2010) Fatty liver disease: the hepatic manifestation of metabolic syndrome. Hypertens Res 33:546–547
Article Google Scholar
- 7.
Guillot X, Semerano L, Saidenberg-Kermanac'h N, Falgarone G, Boissier M-C (2010) Vitamin D and inflammation. Jt Bone Spine 77:552–557
CAS Article Google Scholar
- 8.
Kitson MT, Roberts SK (2012) D-livering the message: the importance of vitamin D status in chronic liver disease. J Hepatol 57:897–909
CAS Article Google Scholar
- 9.
Kwok RM, Torres DM, Harrison SA (2013) Vitamin D and nonalcoholic fatty liver disease (NAFLD): is it more than just an association? Hepatology 58:1166–1174
CAS Article Google Scholar
- 10.
Abramovitch S, Dahan-Bachar L, Sharvit E, Weisman Y, Tov AB, Brazowski E, Reif S (2011) Vitamin D inhibits proliferation and profibrotic marker expression in hepatic stellate cells and decreases thioacetamide-induced liver fibrosis in rats. Gut 60:1728–1737
CAS Article Google Scholar
- 11.
Zhou QG, Hou FF, Guo ZJ, Liang M, Wang GB, Zhang X (2008) 1,25-Dihydroxyvitamin D improved the free fatty-acid-induced insulin resistance in cultured C2C12 cells. Diabetes Metab Res Rev 24:459–464
CAS Article Google Scholar
- 12.
Konstantakis C (2016). Vitamin D deficiency in patients with liver cirrhosis. Ann Gastroenterol.
- 13.
Liangpunsakul S, Chalasani N (2011) Serum vitamin D concentrations and unexplained elevation in ALT among US adults. Dig Dis Sci 56:2124–2129
CAS Article Google Scholar
- 14.
Holick MF (2006) High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc 81:353–373
CAS Article Google Scholar
- 15.
Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B (2006) Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr 84:18–28
CAS Article Google Scholar
- 16.
Dawson-Hughes B, Heaney RP, Holick MF, Lips P, Meunier PJ, Vieth R (2005) Estimates of optimal vitamin D status. Osteoporos Int 16:713–716
CAS Article Google Scholar
- 17.
Yousif MM, Sadek AMEM, Farrag HA, Selim FO, Hamed EF, Salama RI (2019) Associated vitamin D deficiency is a risk factor for the complication of HCV-related liver cirrhosis including hepatic encephalopathy and spontaneous bacterial peritonitis. Intern Emerg Med 14:753–761
Article Google Scholar
- 18.
Angulo P, Hui JM, Marchesini G, Bugianesi E, George J, Farrell GC, Enders F, Saksena S, Burt AD, Bida JP, Lindor K, Sanderson SO, Lenzi M, Adams LA, Kench J, Therneau TM, Day CP (2007) The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology 45:846–854
CAS Article Google Scholar
- 19.
Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J, Sulkowski MS, Torriani FJ, Dieterich DT, Thomas DL, Messinger D, Nelson M (2006) Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 43:1317–1325
CAS Article Google Scholar
- 20.
Das C, Baruah M, Singh D (2013) Imaging of non alcoholic fatty liver disease: A road less travelled. Indian J Endocrinol Metab 17:990
Article Google Scholar
- 21.
Arteh J, Narra S, Nair S (2010) Prevalence of vitamin D deficiency in chronic liver disease. Dig Dis Sci 55:2624–2628
CAS Article Google Scholar
- 22.
Lange CM, Bojunga J, Ramos-Lopez E, von Wagner M, Hassler A, Vermehren J, Herrmann E, Badenhoop K, Zeuzem S, Sarrazin C (2011) Vitamin D deficiency and a CYP27B1-1260 promoter polymorphism are associated with chronic hepatitis C and poor response to interferon-alfa based therapy. J Hepatol 54:887–893
CAS Article Google Scholar
- 23.
Pacifico L, Osborn JF, Bonci E, Pierimarchi P, Chiesa C (2019) Association between vitamin D levels and nonalcoholic fatty liver disease: potential confounding variables. Mini-Reviews Med Chem 19:310–332
CAS Article Google Scholar
- 24.
Jaruvongvanich V, Ahuja W, Sanguankeo A, Wijarnpreecha K, Upala S (2017) Vitamin D and histologic severity of nonalcoholic fatty liver disease: a systematic review and meta-analysis. Dig Liver Dis 49:618–622
CAS Article Google Scholar
- 25.
Targher G, Bertolini L, Scala L, Cigolini M, Zenari L, Falezza G, Arcaro G (2007) Associations between serum 25-hydroxyvitamin D3 concentrations and liver histology in patients with non-alcoholic fatty liver disease. Nutr Metab Cardiovasc Dis 17:517–524
CAS Article Google Scholar
- 26.
Ehrampoush E, Ghaemi A, Osati S, Maleki M, Raeisi Shahraki H, Honarvar B, Bahmanyar M, Homayounfar R (2019) The association between serum vitamin D level and nonalcoholic fatty liver disease. Hepat Mon 19(9):e92992
Article Google Scholar
- 27.
Ma X, Liu S, Zhang J, Dong M, Wang Y, Wang M, Xin Y (2020) Proportion of NAFLD patients with normal ALT value in overall NAFLD patients: a systematic review and meta-analysis. BMC Gastroenterol 14(1):20
Google Scholar
- 28.
Bhatt SP, Nigam P, Misra A, Guleria R, Qadar Pasha MA (2013) Independent associations of low 25 hydroxy vitamin D and high parathyroid hormonal levels with nonalcoholic fatty liver disease in Asian Indians residing in north India. Atherosclerosis 230:157–163
CAS Article Google Scholar
- 29.
Black LJ, Jacoby P, She Ping-Delfos WC, Mori TA, Beilin LJ, Olynyk JK, Ayonrinde OT, Huang RC, Holt PG, Hart PH, Oddy WH, Adams LA (2014) Low serum 25-hydroxyvitamin D concentrations associate with non-alcoholic fatty liver disease in adolescents independent of adiposity. J Gastroenterol Hepatol 29:1215–1222
CAS Article Google Scholar
- 30.
Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF (2000) Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 72:690–693
CAS Article Google Scholar
- 31.
Manco M, Ciampalini P, Nobili V (2010) Low levels of 25-hydroxyvitamin D3 in children with biopsy-proven nonalcoholic fatty liver disease. Hepatology 51:2229–2229
Article Google Scholar
Download references
Acknowledgements
Authors would like to thank Dr. Hussien Ahmed for the great help in statistics and editorial support. All patients were acknowledged for participations in this research.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethics declarations
Ethics approval and consent to participate
The institutional review board approved the study (ZU-IRB#3776-30-5-2017). Written informed consent was obtained from all individual participants in the study.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this article
Gad, A.I., Elmedames, M.R., Abdelhai, A.R. et al. The association between vitamin D status and non-alcoholic fatty liver disease in adults: a hospital-based study. Egypt Liver Journal 10, 25 (2020). https://doi.org/10.1186/s43066-020-00033-z
Download citation
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s43066-020-00033-z
Keywords
- Non-alcoholic fatty liver disease
- Vitamin D
- Risk factors
Can Low Vitamin D Cause Fatty Liver
Source: https://eglj.springeropen.com/articles/10.1186/s43066-020-00033-z



0 Komentar